|
10/27/2022 0 Comments Carbon dioxide poisoning  As a reference, OSHA (Occupational Safety and Health Administration) has set a CO2 permissible exposure limit (PEL) of 5,000 ppm over an eight-hour period and 30,000 ppm over a 10-minute period.Ĭarbon monoxide is a far more dangerous gas. At 80,000 ppm, CO2 can be life-threatening. Symptoms of mild carbon dioxide poisoning include headaches and dizziness at concentrations less than 30,000 ppm. Excess carbon dioxide uses up space in the air instead of oxygen, creating an environment for asphyxiation. While carbon dioxide poisoning is rare, a high concentration of it in a confined space can be toxic. 
Health Risks for Carbon Monoxide and Carbon Dioxide While carbon monoxide is usually an unwanted by-product, packaged carbon monoxide is used in a variety of industries including metal fabrication, chemical manufacturing, pharmaceuticals, electronic and semiconductor applications, and for reducing ores when manufacturing metal carbonyls. Workers near or within enclosed areas or confined spaces such as manholes, garages, tunnels, loading docks, warehouses, vehicle repair shops, and splicing vehicles are also at risk. Truck drivers, forklift operators, or people working near this type of equipment are at a higher risk of exposure. 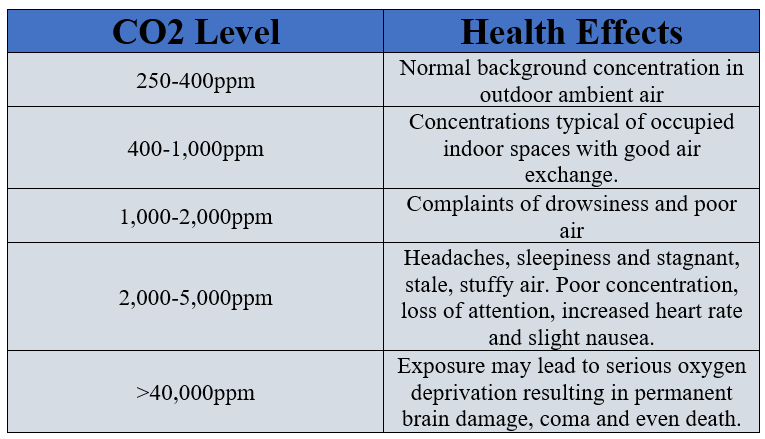
Many furnaces and ovens produce large amounts of the gas, especially when they are not maintained properly. In industrial settings, the internal combustion engine is the chief source of carbon monoxide. 
As a colorless and odorless gas, you can’t identify it without gas detectors. Low levels of oxygen and low temperatures lead to carbon monoxide in the combustion mixture.ĭangerous levels of carbon monoxide can be produced by any fuel-burning appliance, including gas furnaces, gas stoves, gas dryers, gas water heaters, fireplaces, and cars. It is created through the incomplete combustion of coal, natural gas, and oil. Unlike carbon dioxide, carbon monoxide does not occur naturally in the atmosphere. Incomplete combustion happens when there is a limited supply of air, so only half as much oxygen adds to the carbon, forming carbon monoxide (CO = one oxygen atom, CO2 = two oxygen atoms). Where does carbon monoxide come from?Ĭarbon monoxide, on the other hand, is the result of incomplete combustion. Carbon dioxide has many applications in the food and beverage industry, including carbonating drinks. Industrial plants that produce hydrogen or ammonia from natural gas, coal, or large-volume fermentation operations are some of the largest commercial producers of carbon dioxide. It is a largely non-reactive gas, and once released, it quickly mixes throughout the atmosphere.Ĭarbon dioxide is also produced through industrial processes. The carbon dioxide is released into the air as a colorless and odorless gas. You’re witnessing complete combustion when you watch a candle burn: candle wax is a hydrocarbon, which reacts with oxygen in the air and the heat from the lit wick. Complete combustion often (but not always) involves a flame. Complete combustion is a chemical reaction in which a hydrocarbon reacts with oxygen to produce carbon dioxide and water. Where does carbon dioxide come from?Ĭarbon dioxide is the result of complete combustion. But these gases are created through different chemical reactions. #Carbon dioxide poisoning how to#You’re probably aware that they are two different gases-but which one is the good one and which is the bad one? Is it even correct to classify them that way? Before getting into how and where carbon monoxide (CO) and carbon dioxide (CO2) affect people and the environment and how to test for them, let’s get a basic understanding of where they come from.īoth gases are a combination of carbon and oxygen, which is why their names are similar. What is the difference between carbon monoxide and carbon dioxide? If you confuse the two, you’re not alone. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
